Recherche translationnelle en santé,
technologie pour la santé et recherche clinique
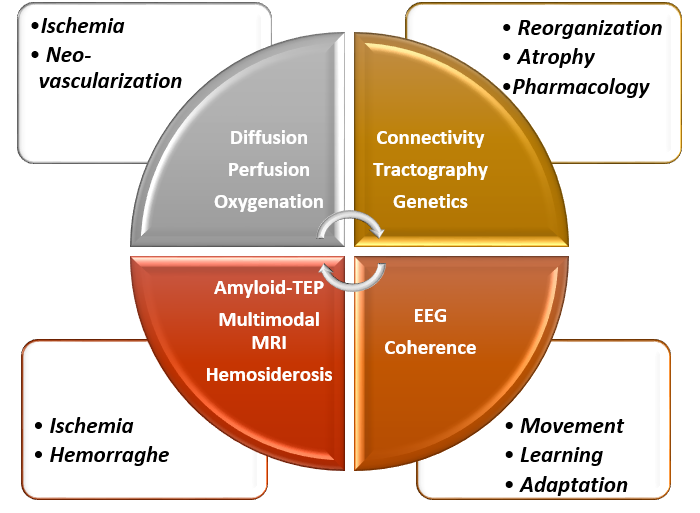
iDREAM team / Axis 2 : Multi-modal imaging
Multi-modal imaging, Post-lesional signatures and predictors
The clinical axis is led by Nicolas Raposo.
The preclinical axis led by Franck Desmoulin.
Scientific objectives
Our objectives are to better understand the physiopathological mechanisms of the neurological disorders and handicaps that we are studying (stroke, head trauma, post-traumatic stress disorder), so as to improve their early diagnosis and to define adapted therapeutic or rehabilitation approaches.
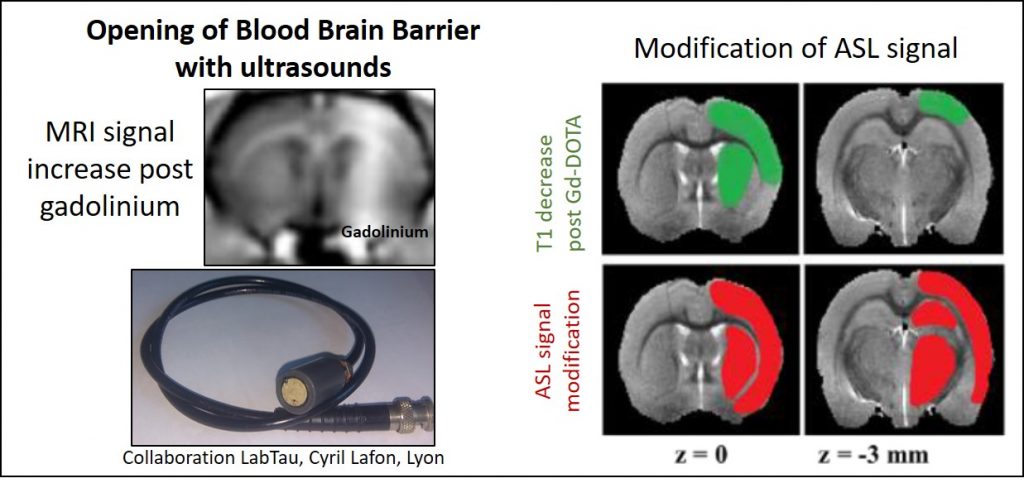
NeuroImaging allows us to define brain targets for our therapies as well as intermediate criteria for the effectiveness of therapeutic approaches.
Clinical imaging
Hemorrhage, MRI and PET
Objectives
In a prospective study of patients with CAA-related lobar intracerebral hemorrhage, we hope to evaluate the role of cortical hemosiderosis in intracerebral hemorrhage recurrence and its interactions with other risk factors for recurrence.
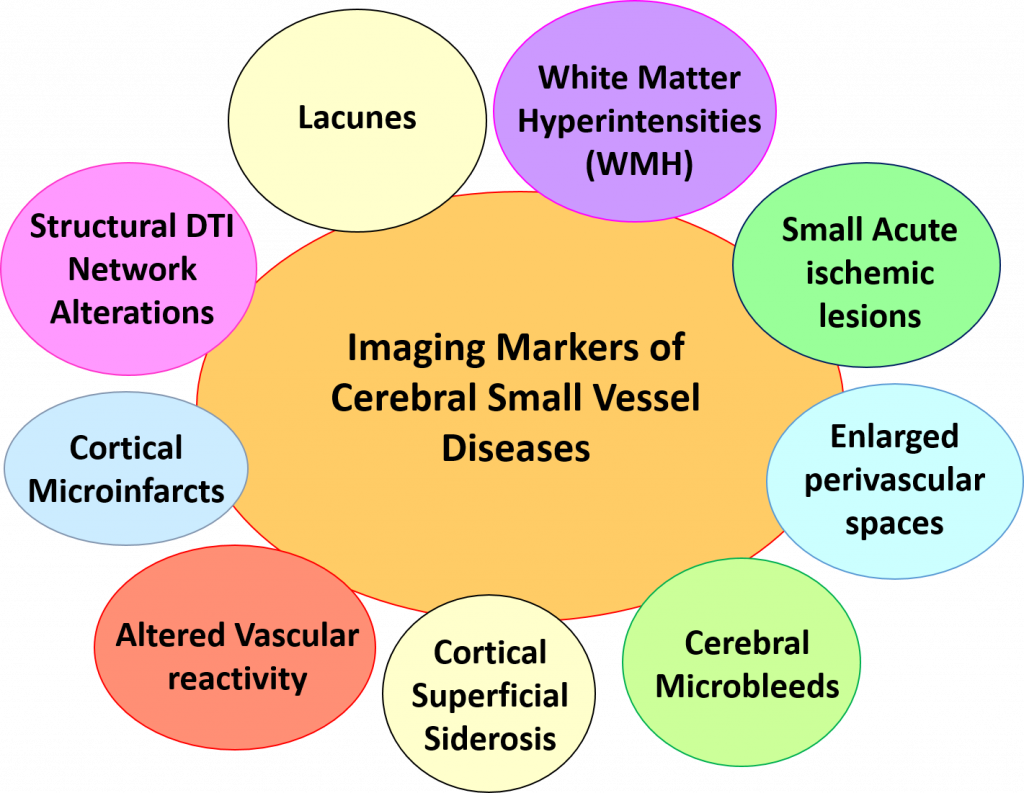
Cerebral small vessel diseases, particularly cerebral amyloid angiopathy (CAA) is one of the main research topics in our group. CAA is a major cause of intracerebral hemorrhage and dementia. Despite important advances in the underlying mechanisms of the disease, no specific treatment is currently available. We aim to develop and evaluate imaging biomarkers of CAA that may have several potential applications as a diagnostic tool and/or prognostic marker for future clinical trials. Several markers have already been highlighted (Raposo et al., Neurology 2017; Eur J Neurology 2017 ; Neurology 2020 ; Am J Neuroradiol 2021).
On going studies
We are coordinating a prospective multicenter study (Toulouse, Bordeaux, Montpellier, Lariboisière) to assess clinical, imaging and genetic factors associated with recurrent intracerebral hemorrhage and dementia following symptomatic intracerebral hemorrhage associated with CAA (Corelia study; clinical trial.org NCT03464344, PI : Nicolas Raposo).
Thrombectomy and MRI
The clinical trial FRAME investigated the yield of the imaging of the ischemic penumbra in patients treated by mechanical thrombectomy during the first 6 hours after onset a time window where multimodal imaging is considered as not necessary to select patients for treatment.
220 patients were included. Our results demonstrate that a substantial number of patients (20%) do not have any more salvageable tissue. In this subgroup, the occurrence of a reperfusion after mechanical thrombectomy (MT) is not associated with a higher rate of neurological recovery, questioning the indication of mechanical thrombectomy in this subgroup. On another hand, 71% of the patients with a significant ischemic penumbra on multimodal imaging who experienced a reperfusion after MT had a significantly higher rate of clinical recovery in comparison with the group of patients who did not; suggesting that they should be the target population for the future development of new revascularization strategies.
In conclusion multimodal imaging performed during the first 6 hours after stroke onset identifies the patients more likely to benefit from mechanical thrombectomy (Olivot et al., Stroke 2021 ; Ann Neurol 2021).
Cerebral palsy and Electroencephalography
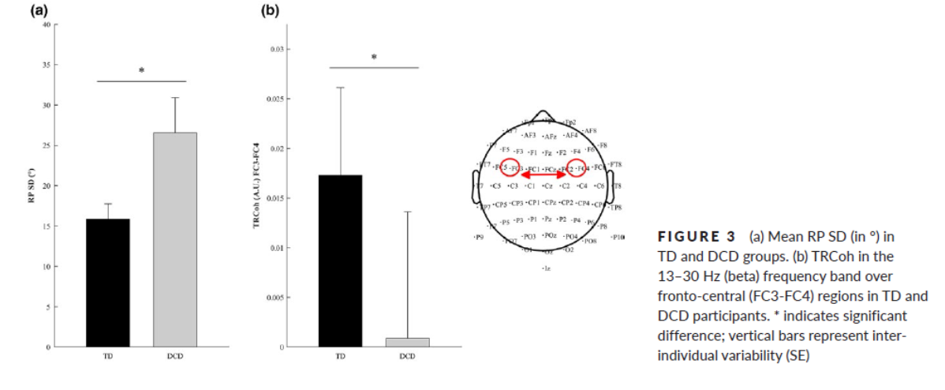
Jessica Tallet and co-workers recorded EEG in children with development coordination disorders (DCD) and controls with typical development (TD). They demonstrated that atypical inter-hemispheric communication correlates with altered motor inhibition during learning of a new bimanual coordination pattern in developmental coordination disorder. Indeed, appropriate motor control involves inhibition of mirror movements, which relies on inter-hemispheric connectivity and coherence (RP: relative phase; TRCoh: task-related coherence) (Blais et al., Developmental Science, 2018).
Stroke and functional connectivity
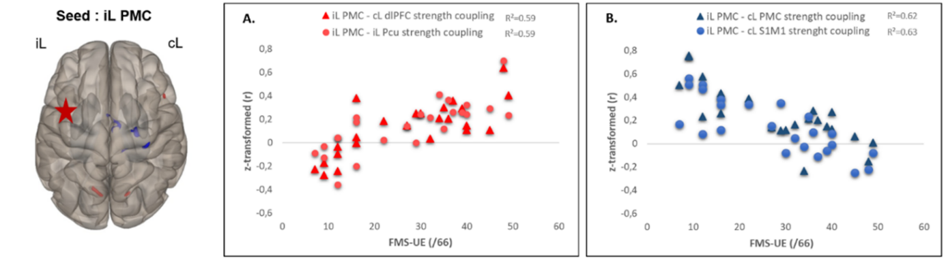
MRI functional connectivity during a sensitivomotor task was investigated in 25 stroke patients and was correlated with motor deficit (Fugl-Meyer Scale of upper limb, FMS-UE). Negative connectivity of ipsilesional premotor cortex with contralesional motor regions (blue clusters) is observed in very impaired patients and evidences disconnection. A cross-modal functional connectivity of the ipsilesional premotor cortex with non-motor regions (prefrontal cortex, precuneus) ws evidenced and may reflect efficient cross-modal compensation strategies (red clusters) (Brihmat et al., Brain Connectivity 2020).
Preclinical imaging
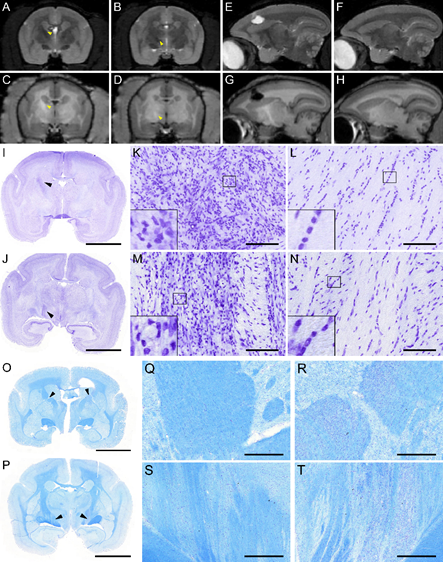
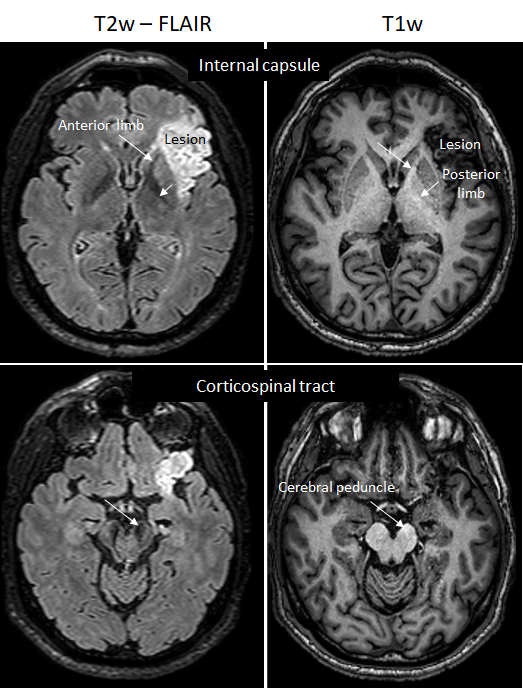
Secondary degeneration biomarker
We have discovered a new biomarker of gliosis accompanying axonal degeneration in stroke patients and in brain-lesioned non-human primates (marmoset). For several months after the cortical lesion, the corticospinal tract undergoes a loss of myelin, an astrocytic infiltration and a microglial reaction. They induce a loss of intensity on T2 MRI scans and a slight hyperintensity on T1 MRI scans (Le Friec et al., Trans Stroke Res 2020).